Bromine-substituted triphenylamine derivatives with improved hole-mobility for highly efficient green phosphorescent OLEDs with a low operating voltage†
Abstract
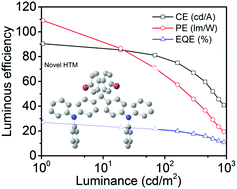
Charge transport materials play a crucial role in organic light-emitting diodes (OLEDs) due to their effect on reducing the operating voltage and enhancing the power efficiency. To develop hole transport materials with improved hole-mobility, two novel bromine-substituted triphenylamine derivatives: ((4-bromo-phenyl)-bis-(4-quinolin-8-yl-phenyl)-amine) Br-DQTPA and (9,9-bis-(4-triphenyl-amine)-2,7-dibromo-9H-fluorene) Br-DTF were designed and synthesized. The hole mobilities of Br-DQTPA and Br-DTF show one order of magnitude enhancement compared with non-brominated (phenyl-bis-(4-quinolin-8-yl-phenyl)-amine) DQTPA and (9,9-bis-(4-triphenyl-amine)-9H-fluorene) DTF under the same electric field. Attributed to their improved hole mobilities, traditional green phosphorescent OLEDs that use Br-DQTPA or Br-DTF as a hole transport material both show an ultralow operating voltage at 1 cd m−2 (below 2.4 V) and very high luminous efficiencies (over 21% and 90 lm W−1) without light outcoupling improvement. Those results are much better than those of DQTPA-, DTF-, and even NPB-based green devices, indicating that bromine-substitution is a promising and convenient way to achieve novel hole transport materials with improved hole-mobility.

 Please wait while we load your content...
Please wait while we load your content...