Full color control and white emission from CaZnOS:Ce3+,Na+,Mn2+ phosphors via energy transfer
Abstract
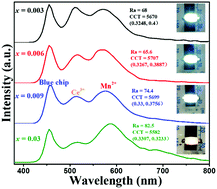
A series of CaZnOS:Ce3+,Na+,Mn2+ phosphors were successfully synthesized by a conventional solid-state reaction and their luminescence properties were investigated in detail. The phosphors can be excited by blue light at 455 nm, matching well with the commercial blue light emitting diode (LED) chips. Tunable full-color emission can be realized by combining the blue chips (455 nm), yellow emission (about 530 nm) ascribed to Ce3+ and red emission (about 590 nm) originating from Mn2+ in the CaZnOS host. Energy transfer (ET) from Ce3+ to Mn2+ in the CaZnOS host was validated by the variation of emission intensities as well as the decay lifetimes of Ce3+ ions with increasing Mn2+ concentration, and the ET mechanism is analyzed and ascribed to be a quadrupole–quadrupole interaction. The composition-controlled CaZnOS:0.03Ce3+,0.03Na+,0.03Mn2+ phosphor shows improved chromaticity characteristics fabricated using a blue LED chip, with an Ra of 82.5, a CCT of 5582 K and color coordinates (0.33, 0.32) at the forward current of 30 mA. We demonstrate that CaZnOS:Ce3+,Na+,Mn2+ show potential applications as a single-component white-light emission phosphor for fabricating white LED devices.

 Please wait while we load your content...
Please wait while we load your content...