Characterization of mesoporous calcium phosphates from calcareous marine sediments containing Si, Sr and Zn for bone tissue engineering†
Abstract
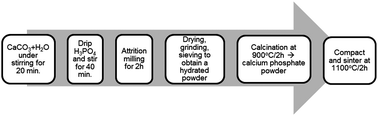
Calcium phosphates (CAPs) can be produced from either biologically sourced materials or mineral deposits. The raw materials impart unique properties to the CAPs due to innate trace amounts of elements that affect the crystal structure, morphology and stoichiometry. Using calcium carbonate (CaCO3) precursors derived from fossilized calcareous marine sediments (FCMSs), we have synthesized a novel class of CAP biomaterials, termed fm-CaPs, with defined Ca/P molar ratios of 1.4 and 1.7 using a wet synthesis method. Compared with commercially available CAP biomaterials, such as hydroxyapatite (HA) and beta-tricalcium phosphate (β-TCP), fm-CaP1.7 had a biphasic composition consisting of an HA phase (in a hexagonal system) and a β-TCP phase (in a rhombohedral crystalline system), which is desirable for the current design of bone substitutes, whereas fm-CaP1.4 consisted of an HA phase and a beta-dicalcium pyrophosphate phase (in a tetragonal system). These bioceramics exhibited a fringe structure of regular crystallographic orientation with well-ordered mesoporous channels. The FCMS raw material imparted trace amounts of silicon (Si), strontium (Sr) and zinc (Zn) to fm-CaPs; these are elements that are important for bone formation. The cyto-compatibility of these biomaterials and their effects on cellular activity were evaluated using osteoblast cells. Cell proliferation assays revealed no signs of cytotoxicity, whereas cells growth was equal to or better than HA and β-TCP controls. The SEM analysis of the cell and material interactions showed good cell spreading on the fm-CaP materials that was comparable to β-TCP and in vitro assays suggested robust osteogenic differentiation, as seen by increased mineralization (alizarin red) and upregulation of osteogenic gene expression. Our results indicate that fm-CaP1.7, in particular, has chemical, physical and morphological properties that make this material suitable for applications that promote bone tissue regeneration.

 Please wait while we load your content...
Please wait while we load your content...