Spheroid culture of primary hepatocytes with short fibers as a predictable in vitro model for drug screening†
Abstract
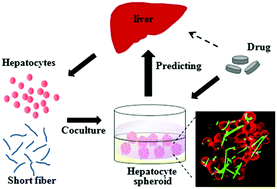
The establishment of a reliable in vitro liver model for drug screening remains challenging with respect to tethering the growth of hepatocyte spheroids and adapting to the current high-throughput system. In the current study, short fibers are utilized as scaffolds for the generation of size-controlled hepatocyte spheroids that recapitulate in vivo hepatic phenotypes and functions. The spheroid formation is modulated by the length and galactose/RGD grafts of short fibers, and short 50 μm long fibers motivate the spheroid formation with optimal hepatic function. Short fibers distribute throughout the entire spheroid for tethering hepatocyte growth to form compact spheroids. Compared with scaffold-free spheroid culture on agarose-coated plates, the spheroid culture with short fibers achieves higher clearance rates of model drugs and provides a better prediction of the in vivo drug clearance rate with a correlation value of 0.886. In addition, the drug metabolism capability is highly sensitive to the inducers and inhibitors of metabolizing enzymes, and the responsiveness is maintained during 20 days of culture, exhibiting an efficient in vitro model for determining drug–drug interactions. Therefore, the spheroid culture with short fibers provides an easily manipulated strategy to maintain hepatocyte functions for a prolonged period and enable ready deployment in conventional multiwell plates and diverse organ-on-a-chip devices for high-throughput drug screening.

 Please wait while we load your content...
Please wait while we load your content...