HP-DO3A-based amphiphilic MRI contrast agents and relaxation enhancement through their assembly with polyelectrolytes†
Abstract
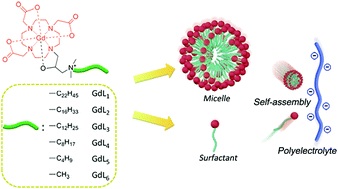
HP-DO3A-based cationic amphiphilic Gd(III) complexes bearing quaternary ammonium salts of different alkyl chain lengths were synthesized. The structures of the intermediates, the ligands and the Gd(III) complexes were characterized by 1H NMR, 13C NMR, ESI and MALDI-TOF mass spectrometry. Relaxation studies of the amphiphilic Gd(III) complexes revealed that long alkyl chains facilitated the formation of micelles in an aqueous solution and afforded high relaxivity. Electrostatic self-assembly of the GdL1 complex with polyacrylate sodium (PAAS) showed significant relaxation enhancement with the relaxivity being up to 20.03 mM−1 s−1 at 1.5 T, which was about 4 times that of the commercial MRI contrast agent, Gd-DTPA. The long hydrophobic chain was found to play an important role in the formation of the self-assembly. Zeta potential and dynamic light scattering measurements were carried out to reveal the aggregation process during the assembly. Good biocompatibility of the complexes and high cell viability were found in HepG-2 and L-02 cells by the MTT assay. The reactive oxygen species (ROS) generation study further confirmed the biocompatibility. In vivo imaging showed significant contrast enhancement in the liver and bladder regions.


 Please wait while we load your content...
Please wait while we load your content...