Phase-changeable and bubble-releasing implants for highly efficient HIFU-responsive tumor surgery and chemotherapy†
Abstract
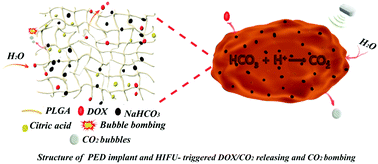
CO2 microbubbles (MBs) were explored as smart agent in suppressing tumors. CO2 MBs can be massively produced from the decomposition of an effervescent disintegrant in water. However, the explosive decomposing of the effervescent disintegrant in water will extremely limit its therapeutic applications. The short retention duration of CO2 MBs in blood and the tiny quality of MBs that can leak into tumor sites also severely decrease the tumor therapy efficiency. In this research, a multifunctional poly(lactic-co-glycolic acid) (PLGA)-based solid implant was facilely in situ constructed within tumors based on the liquid–solid phase transition of hydrophobic PLGA/effervescent disintegrant/doxorubicin (DOX) (denoted as PED) oleosol, to effectively control the decomposition of the effervescent disintegrant and increase the accumulation of MBs in the tumor site. The formed PED implant could simultaneously function as a high intensity focused ultrasound (HIFU) enhancement agent and a drug delivery platform featured with HIFU-responsive DOX and spontaneous MBs releasing characteristics. Without HIFU treatment, the hydrophobic PLGA will prevent the contacting of effervescent disintegrant or DOX with water. Under HIFU treatment, water could easily diffuse into the PED implant and thus DOX release, effervescent disintegrant decomposition and CO2 release will be switched on. These CO2 MBs can oscillate with the external focused ultrasound and enhance the mechanical and cavitation effect of focused ultrasound, resulting in an enhanced tumor HIFU ablation towards a VX2 tumor-bearing New Zealand White rabbit. Importantly, it is difficult for effervescent disintegrant and DOX to enter the bloodstream in quantity, therefore, the utilization of CO2 MBs and DOX can be increased and the in vivo biosafety of the implant can be guaranteed. The developed PED implant features easy fabrication, low cost, and excellent in vivo hemo-/histo-compatibility, and holds promising potential for future clinical translation for localized tumor therapy.


 Please wait while we load your content...
Please wait while we load your content...