A new peptide-based fluorescent probe selective for zinc(ii) and copper(ii)†
Abstract
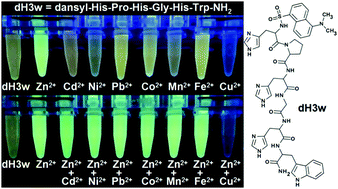
A novel metal ion-sensitive fluorescent peptidyl-probe has been designed based on the most common five-residue repeat in mammalian histidine rich glycoproteins (HRGs). A dansyl-amide moiety at the N-terminus and a tryptophan residue at the C-terminus of the peptide were added as they can act as a FRET (fluorescence resonance energy transfer) pair. The dansyl fluorophore was chosen also because it frequently shows strong CHEF (chelation enhanced fluorescence) and solvatochromic effects. The designed peptide, dansyl-HPHGHW-NH2 (dH3w), showed a selective fluorescence turn-on response to Zn2+ in aqueous solutions at pH 7.0 when excited at both 295 nm and 340 nm, thus indicating that both FRET and CHEF or solvatochromic effects are active in the metal/peptide complex. Steady-state fluorescence and isothermal titration calorimetry (ITC) measurements demonstrated that two peptide molecules bind to one zinc ion with an association constant Ka = 5.7 × 105 M−1 at 25 °C and pH 7.0. The fluorescence response to Zn2+ was not influenced by Pb2+, Cd2+, Mn2+, Fe2+, Fe3+, Mg2+, Ca2+, K+ and Na+ ions and only slightly influenced by Co2+ and Ni2+. Copper(II), at concentrations as low as 5 μM, caused a strong quenching of both free and Zn2+ complexed dH3w. The determination of the binding parameters for Cu2+ has shown that one copper ion binds to one dH3w molecule with an association constant of 1.2 × 106 M−1 thus confirming the higher affinity of peptide for Cu2+ than for Zn2+. Finally, we demonstrated that dH3w can penetrate into HeLa cells and could thus be used for the determination of intracellular Zn2+ and Cu2+ concentrations.


 Please wait while we load your content...
Please wait while we load your content...