Surface engineering on a nanocatalyst: basic zinc salt nanoclusters improve catalytic performances of Ru nanoparticles†
Abstract
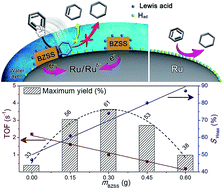
Herein, we report novel surface-modified Ru-based catalysts by the chemisorption of basic zinc sulfate salts (3Zn(OH)2·ZnSO4·xH2O, BZSSs) and demonstrate their enhanced selectivity toward cyclohexene (CHE) in benzene-selective hydrogenation. BZSS nanoclusters are confirmed to regulate the surface and electronic properties of Ru nanoparticles. The surface active sites on Ru nanoparticles are reconstructed because the strong active sites are selectively occupied and blocked by BZSS nanoclusters. Lewis acid active sites, which are introduced by the BZSS and modified by the interaction between Ru(0) and the BZSS, can retain the activity of the Ru catalyst and greatly improve the selectivity toward CHE. Benefiting from the BZSS nanoclusters located on the Ru nanoparticles, the surface-modified catalysts present excellent selectivity with high activity for the hydrogenation reaction. This is particularly clear in that the catalyst operated stably for more than 600 h on an industrial production line; the benzene conversion was maintained at 40%, and the selectivity toward CHE was maintained over 80%.


 Please wait while we load your content...
Please wait while we load your content...