A stability study of polymer solar cells using conjugated polymers with different donor or acceptor side chain patterns†
Abstract
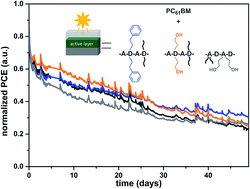
Improvement of the power conversion efficiency and long term stability remains to be of crucial importance for the further development of polymer solar cells (PSCs). Herein, a donor–acceptor copolymer based on 4,8-di(thiophene-2′-yl)benzo[1,2-b:4,5-b′]dithiophene (DTBDT) and 4,7-di(thiophene-2′-yl)benzo[c][1,2,5]thiadiazole (DTBT), specifically selected because of its suitability for roll-coating in the ambient environment, is investigated in terms of operational stability via partial exchange (5 or 10%) of the alkyl side chain on either the donor or the acceptor monomer with a 2-hydroxyethyl or 2-phenylethyl group. It is shown that the exchange of the hexyl chain on the DTBT moiety has a negative impact on the stability of the polymer as well as on the performance of the resulting PSCs. On the other hand, partial exchange of the 2-hexyldecyl side chain of the BDT unit by a 2-hydroxyethyl group results in an improved photochemical stability of the polymer film and a higher efficiency of 5.6% for the spin-coated PSC. The stability of roll-coated devices also slightly increases with the incorporation of 10% of either the 2-hydroxyethyl or 2-phenylethyl side chain.

 Please wait while we load your content...
Please wait while we load your content...