A copper-based sorbent with oxygen-vacancy defects from mechanochemical reduction for carbon disulfide absorption†
Abstract
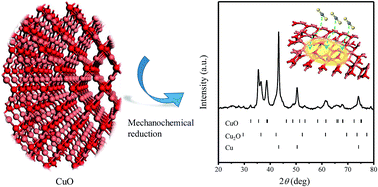
This work innovatively provides an efficient method to prepare a Cu-based sorbent with oxygen-vacancy (Ovac) defects via mechanochemical reduction of CuO for CS2 absorption. Crystalline structures based on XRD measurement show that mechanical force can lead to CuO reduction and Ovac defect generation during ball milling of CuO with Mg as a reducing agent. Sulfur content measurement shows that the prepared Cu-based sorbent with Ovac defects can realize efficient CS2 absorption at 150 °C with a breakthrough sulfur capacity of 10.7 wt%. Electronic structures based on first-principles calculation show that the Ovac defects can decrease copper coordination, increase the surface energy and electron density, and reduce the band gap by forming coordinatively unsaturated Cu, which benefit the chemisorption of CS2 molecules. Cu2O with Ovac defects, the main phase for CS2 absorption in the sorbent, can grab CS2 molecules by the trapping effect of its Ovac defects with a chemisorption distance of 1.53 Å and an energy of −110.45 kJ mol−1, and offer electrons for breaking C–S bonds. This work paves a new pathway for the preparation of Cu-based sorbents with high performance for sulfur compound absorption.

 Please wait while we load your content...
Please wait while we load your content...