Concave octahedral Pd@PdPt electrocatalysts integrating core–shell, alloy and concave structures for high-efficiency oxygen reduction and hydrogen evolution reactions†
Abstract
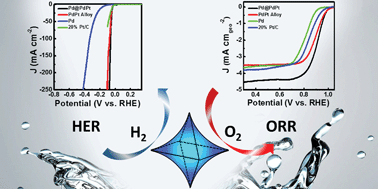
The development of bifunctional catalysts for both the hydrogen evolution reaction (HER) and oxygen reduction reaction (ORR) is crucial for facile hydrogen production via water splitting and reducing oxygen to promote electrochemical energy conversion in fuel cells. Here, we prepare a unique concave octahedral Pd@PdPt electrocatalyst, which integrates three structural types, core–shell, concave and alloy structures, using an ethylene glycol system. Scanning transmission electron microscopy (STEM), energy-dispersive X-ray spectroscopy (EDS) line-scan, and X-ray photoelectron spectroscopy (XPS) analyses reveal that the concave octahedral Pd core is surrounded by a PdPt alloy shell. Through some control experiments, a possible mechanism for the formation of the nanostructure is proposed. The as-prepared Pd@PdPt NCs exhibit a superior enhanced bifunctional electrocatalytic performance for both the ORR and the HER, even better than that of 20% Pt/C. When used in the ORR, the concave octahedral Pd@PdPt NCs exhibit a superior half-potential of 0.91 V (vs. RHE), a large mass activity of 0.95 A mgPt−1, and a superior stability over 1000 cycles in 0.1 M KOH. When used in the HER, these NCs present a positive onset potential of −5 mV (vs. RHE), a small Tafel slope of 38 mV dec−1, a lower overpotential of ∼39 mV at a current density of 10 mA cm−2 and a long-term durability over 4000 cycles in 0.5 M H2SO4. This study enables the design of multi-structural bifunctional electrocatalysts for the renewable energy field.


 Please wait while we load your content...
Please wait while we load your content...