Zn electrode with a layer of nanoparticles for selective electroreduction of CO2 to formate in aqueous solutions†
Abstract
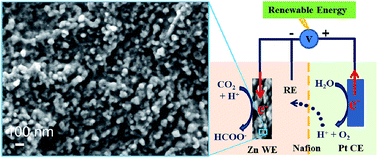
Developing inexpensive and non-toxic electrocatalysts that can reduce CO2 to formate with high selectivity and stability as well as high current densities is an important step for a sustainable carbon cycle. Electrocatalysts that show the highest faradaic efficiency for formate, such as Pb and Hg, are toxic or expensive and have low current densities. Bulk Zn is a cheap metal that has historically been identified to be a CO2 to CO conversion catalyst. In this work, we introduce a novel Zn electrode with a layer of nanoparticles that exhibit high performance toward CO2 electrochemical reduction to produce formate in aqueous solution. The maximum faradaic efficiency for formate production of over 87% is achieved with a formate partial current density of 12.8 mA cm−2, which are almost 8 times higher than that of bulk Zn foil and 17 times higher than that of bulk Zn foil, respectively. The improvement in catalytic performance is attributed to the catalytically active facets and special surface structure of polycrystalline Zn formed during reduction of polycrystalline ZnO. The catalyst shows no obvious performance deterioration during the 14 h continuous CO2 reduction.

 Please wait while we load your content...
Please wait while we load your content...