Fabrication of metallic charge transfer channel between photoanode Ti/Fe2O3 and cocatalyst CoOx: an effective strategy for promoting photoelectrochemical water oxidation†
Abstract
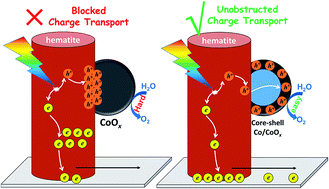
Cocatalysts are one essential factor for promoting the photoelectrochemical water oxidation efficiency of photoanodes, but the influence of interface charge transfer between the semiconductor and cocatalyst has been rarely addressed. Due to their poor charge conductivity, the application of many metal oxides as cocatalysts has been limited. In this work, using photoanode Ti/Fe2O3 and cocatalyst CoOx as a model system, we elaborately design a charge transfer channel of metal Co between the photoanode and cocatalyst. Such a ternary nanostructure has proved to possess several advantageous features, including effective charge separation efficiency, long lifetime of photogenerated holes, and fast charge transport from the photoanode to the surface of the cocatalyst. Benefiting from these desirable nanostructures, the photoanode, containing a charge transfer channel of metal Co, exhibited a remarkable photoelectrochemical performance for water oxidation, with an almost 7-fold enhancement in the photocurrent density at 1.23 V vs. RHE under visible light illumination and about a 0.15 V water oxidation onset potential negative shift compared to the Ti/Fe2O3 photoanode. These findings demonstrate that the successful construction of a metallic charge transfer channel between the photoanode and cocatalyst should be employed as a general strategy for photoconversion applications.

 Please wait while we load your content...
Please wait while we load your content...