Inexpensive electrochemical synthesis of nickel iron sulphides on nickel foam: super active and ultra-durable electrocatalysts for alkaline electrolyte membrane water electrolysis†
Abstract
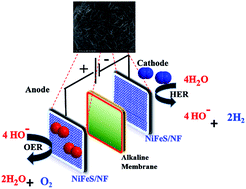
Efficient and durable electrocatalysts for water splitting (both the oxygen evolution reaction (OER) and hydrogen evolution reaction (HER)) are required for the replacement of expensive noble metal-based catalysts used in water electrolysis devices. Herein, we describe the inexpensive synthesis of electrochemically deposited nickel iron sulphides on nickel foam (NiFeS/NF) and their use as highly efficient bi-functional water splitting catalysts that can meet the current energy demands. The NiFeS/NF electrocatalysts exhibited low overpotentials of approximately 231 mV for the OER and 180 mV for the HER in 1 M KOH. Moreover, the NiFeS/NF electrocatalysts exhibited a turnover frequency of one order of magnitude higher than those of state-of-the-art noble metal catalysts (RuO2 and Pt/C) for the OER and HER. In addition to this excellent activity, the bi-functional catalyst also exhibited an outstanding OER and HER electrode stability for over 200 h with minimal loss in activity. Further, it can be directly used in the practical application of alkaline electrolyte membrane water electrolysis. The high performance, prolonged electrode stability, and facile synthesis as an adherent coating on nickel foam suggest that NiFeS/NF electrocatalysts might be suitable for use as alternative commercially viable catalysts.


 Please wait while we load your content...
Please wait while we load your content...