Doped graphenes as anodes with large capacity for lithium-ion batteries†
Abstract
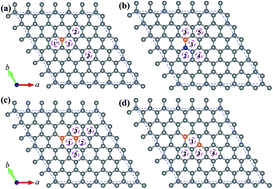
To understand the chemical doping effect on the lithium (Li) storage of graphene, we have performed first-principles calculations to study the adsorption and diffusion of Li adatoms on boron (B)- and nitrogen (N)-doped graphenes, which include individual and paired B (and N) dopants in graphene. Our results show that the adsorption of a Li adatom on B-doped graphene is exothermic, in contrast to a more endothermic adsorption on graphitic N doped graphene. Particularly on the meta B–B pair doped graphene the adsorption energy of a Li adatom is lowered by about 1.84 eV with respect to that on the un-doped graphene. On B-doped graphene, Li adatoms would tend to migrate toward the B-doped region and unfavorably migrate outward. Based on the stable structures of multiple Li adatoms on graphene, which are identified by globally searching with the assistance of the particle swarm optimization (PSO) algorithm, we have analysed the Li adsorption and the electronic structures of graphene substrates via the states-filling model. The B-doped graphenes would achieve a maximum Li capacity of Li6.84B2C70, demonstrating a significant enhancement of Li capacity for graphene as the anode material of LIBs.

 Please wait while we load your content...
Please wait while we load your content...