Sulfur rich electron donors – formation of singlet versus triplet radical ion pair states featuring different lifetimes in the same conjugate†
Abstract
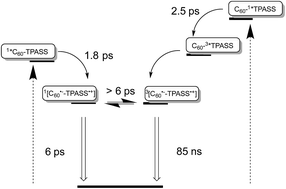
An unprecedented family of novel electron-donor acceptor conjugates based on fullerenes as electron acceptors, on one hand, and triphenyl amines as electron donors, on the other hand, have been synthesized and characterized in a variety of solvents using steady state absorption/emission as well as transient absorption spectroscopy. These are unprecedented in terms of their outcome of radical ion pair formation, that is, the singlet versus triplet excited state. This was corroborated by femto/nanosecond pump probe experiments and by molecular orbital calculations. Not only has the donor strength of the triphenylamines been systematically altered by appending one or two sulfur rich dithiafulvenes, but the presence of the latter changed the nature of the radical ion pair state. Importantly, depending on the excitation wavelength, that is, either where the fullerenes or where the triphenylamines absorb, short-lived or long-lived radical ion pair states, respectively, are formed. The short-lived component with a lifetime as short as 6 ps has singlet character and stems from a fullerene singlet excited state precursor. In contrast, the long-lived component has a lifetime of up to 130 ns in THF, has triplet character, and evolves from a triplet excited state precursor. Key in forming more than three orders of magnitude longer lived radical ion pair states is the presence of sulfur atoms, which enhance spin–orbit coupling and, in turn, intersystem crossing. Independent confirmation for the singlet versus triplet character came from temperature dependent measurements with a focus on the radical ion pair state lifetimes. Here, activation barriers of 2.4 and 10.0 kJ mol−1 for the singlet and triplet radical ion pair state, respectively, were established.

 Please wait while we load your content...
Please wait while we load your content...