Structural changes correlated with magnetic spin state isomorphism in the S2 state of the Mn4CaO5 cluster in the oxygen-evolving complex of photosystem II†
Abstract
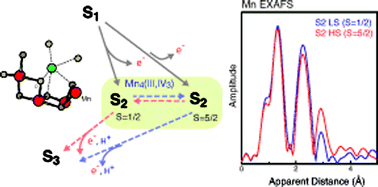
The Mn4CaO5 cluster in photosystem II catalyzes the four-electron redox reaction of water oxidation in natural photosynthesis. This catalytic reaction cycles through four intermediate states (Si, i = 0 to 4), involving changes in the redox state of the four Mn atoms in the cluster. Recent studies suggest the presence and importance of isomorphous structures within the same redox/intermediate S-state. It is highly likely that geometric and electronic structural flexibility play a role in the catalytic mechanism. Among the catalytic intermediates that have been identified experimentally thus far, there is clear evidence of such isomorphism in the S2 state, with a high-spin (5/2) (HS) and a low spin (1/2) (LS) form, identified and characterized by their distinct electron paramagnetic resonance (EPR spectroscopy) signals. We studied these two S2 isomers with Mn extended X-ray absorption fine structure (EXAFS) and absorption and emission spectroscopy (XANES/XES) to characterize the structural and electronic structural properties. The geometric and electronic structure of the HS and LS S2 states are different as determined using Mn EXAFS and XANES/XES, respectively. The Mn K-edge XANES and XES for the HS form are different from the LS and indicate a slightly lower positive charge on the Mn atoms compared to the LS form. Based on the EXAFS results which are clearly different, we propose possible structural differences between the two spin states. Such structural and magnetic redox-isomers if present at room temperature, will likely play a role in the mechanism for water-exchange/oxidation in photosynthesis.

 Please wait while we load your content...
Please wait while we load your content...