Synthesis and characterization of lipophilic cationic Ga(iii) complexes based on the H2CHXdedpa and H2dedpa ligands and their 67/68Ga radiolabeling studies†
Abstract
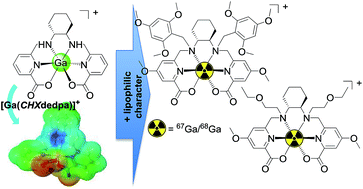
68 Ga is an attractive isotope for incorporation into a positron-emission tomography (PET) imaging agent, and is finding use as an alternative generator-produced isotope to 99mTc particularly in imaging of myocardial perfusion. We have synthesized six new chelating ligands based on our previously reported H2dedpa and H2CHXdedpa scaffolds (CHX = cyclohexyl, H2dedpa = 1,2-[[carboxypyridin-2-yl]methylamino]ethane). These ligands are designed to incorporate several lipophilic appendages at the secondary nitrogens, and upon coordination to 68Ga(III) will form lipophilic, cationic complexes designed to mimic the properties of other clinically relevant myocardial perfusion imaging agents. The non-radioactive Ga(III) complexes were prepared and characterized by NMR spectroscopy; each ligand retained its predicted hexadentate N4O2 binding to Ga(III). The radiolabeling properties of the six ligands were assessed using the longer-lived 68Ga surrogate, 67Ga. The absence of ‘free’ uncomplexed 67Ga in the HPLC radio-chromatograms indicated >99% radiochemical yields (10 minutes at ambient temperature, ligand concentrations of 10−4 M). However, the N,N′-benzyl functionalized derivatives displayed multiple peaks corresponding to the presence of additional 67Ga-complexes which complicated further study. Selected 67Ga-CHXdedpa complexes were tested for in vitro stability against the metal-binding protein apo-transferrin, and were found to be sufficiently stable (>80%) in a 2 h challenge assay, suggesting that alternative N,N′-alkylated derivatives which introduce more lipophilic character will be of interest in future studies.


 Please wait while we load your content...
Please wait while we load your content...