The synthesis and electrical properties of hybrid gel electrolytes derived from Keggin-type heteropoly acids and 3-(pyridin-1-ium-1-yl)propane-1-sulfonate (PyPs)†
Abstract
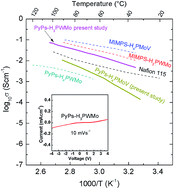
Herein, we report the effect of the proton concentration in polyoxometalates (POMs) upon hybrid formation with ionic liquids (ILs), and their ionic conductivity relationship to optimize their ionic conductivity. The hybrid gels were derived from Keggin-type heteropoly acids containing different proton concentrations, such as H3PW11MoO40, H4PMo11VO40 and H5PMo10V2O40, and 3-(pyridin-1-ium-1-yl)propane-1-sulfonate (PyPs) IL. Elemental C, H, and N analysis was found to be consistent with the theoretical composition within 4% for C and N, whereas H content was found to be slightly higher than the anticipated value, which may be due to potential uptake of water during the sample preparation. 1H and 13C nuclear magnetic resonance and Fourier transform infrared spectroscopy (FTIR) confirmed the presence of functional groups of PyPs in the hybrids. In situ variable temperature powder X-ray diffraction (PXRD), thermogravimetric analysis (TGA), electrochemical AC impedance spectroscopy and cyclic voltammetry studies showed excellent thermal (up to ∼300 °C) and electrochemical (3 V at room temperature) stability of [PyPs]3PW11MoO40. The structural characterizations confirmed the interaction between the organic cation and Keggin-type inorganic heteropoly anion in the hybrid material. The bulk ionic conductivity of 0.1, 0.01 and 0.0003 S cm−1 at ∼90 °C was obtained for [PyPs]3PW11MoO40, [PyPs]4PMo11VO40 and [PyPs]5PMo10V2O40, respectively.

 Please wait while we load your content...
Please wait while we load your content...