Controlling red upconversion luminescence in Gd2O3:Yb3+–Er3+ nanoparticles by changing the different atmosphere†
Abstract
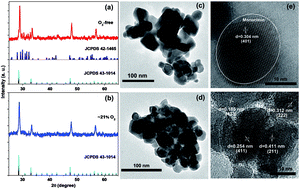
Lanthanide (Ln3+)-doped nanoparticles that produce strong red upconversion (UC) luminescence are desirable for biological applications. For nanoparticles, modification of their electronic properties and stabilization of their crystallographic phase are crucial for tuning their luminescence behaviors. Here, Yb3+–Er3+ doped Gd2O3 nanoparticles have been synthesized by calcining Gd2O2S in nitrogen (O2-free) and ambient air (∼21% O2), respectively. Various approaches including X-ray diffraction (XRD), transmission electron microscopy (TEM), Raman spectroscopy and thermo-gravimetric and differential thermal analyses (TG-DTA) were used to characterize the structure, morphology and phase formation mechanism of the nanoparticles. Upconversion luminescence properties were investigated by emission intensity vs. excitation power (double logarithmic relationship) and temperature dependent emission spectroscopy. The different splitting transition probabilities of the cubic and/or monoclinic phase, the probability of multiphonon relaxations and cross relaxations between Er3+ ions are responsible for the efficient red emission in the cubic Gd2O3 nanoparticles. More importantly, the lower phonon energy of the pure cubic sample has good thermal stability in the current detectable range of 20–100 °C. These results imply that the cubic Gd2O3 upconversion nanoparticles may have multiplexing functionality in bio-imaging.

 Please wait while we load your content...
Please wait while we load your content...