Study on the structural and electrocatalytic properties of Ba2+- and Eu3+-doped silica xerogels as sensory platforms†
Abstract
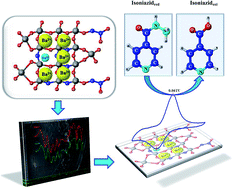
This work reports on the sol–gel synthesis of barium- and europium-doped silica xerogel and its use as an electrocatalytic sensor. Barium was chosen to assist the network framing and europium to provide electronic properties, including the Eu2+/Eu3+ redox process. The results from different molecular and structural techniques indicated that this xerogel is a composite material that combines a non-crystalline polymeric silica network with a high level of agglomeration and a low degree of reticulation formed by the hydrolysis and condensation of TEOS and M–O bonds (M = Ba2+ and/or Eu3+), whereas the oxygen atoms belong to the xerogel phase. The electrochemical behavior of the silica xerogel:Ba2+,Eu3+ with different amounts of Eu3+ under several different conditions was investigated. The electrochemical sensing platform showed a well-defined redox coupling with a formal potential of 0.06 V, assigned to europium redox sites in silicate. Electrocatalytic activity was observed with an increasing anodic peak current to the isoniazid oxidation, indicating that the electrochemical sensing platform designed here was successfully achieved.


 Please wait while we load your content...
Please wait while we load your content...