Utilization of a new gold/Schiff-base iron(iii) complex composite as a highly sensitive voltammetric sensor for determination of epinephrine in the presence of ascorbic acid†
Abstract
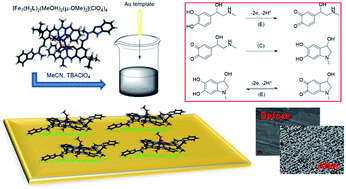
The preparation of new materials that can act as systems capable of sensing biologically relevant molecules constitutes a significant modern challenge as well as a necessity oriented towards disease prevention. Subcomponent self-assembly of 2-(methylhydrazino)benzimidazole, 4-tert-butyl-2,6-diformylphenol and Fe(ClO4)2(H2O)6 leads to a new, bimetallic iron(III) complex of the following formula: [Fe2(H3L)2(MeOH)2(μ-OMe)2](ClO4)4 (1), as established by ESI-MS, FTIR and single crystal X-ray analysis. It is important to note that ligand H3L was also successfully synthesized and characterized for the first time. Compound 1 was successfully deposited on a gold electrode and applied as a voltammetric sensor with respect to epinephrine (EP). Cyclic voltammograms (CVs) proved the catalytic activity of the new, electrochemically prepared composite Au/1 for the oxidation of EP in the presence of ascorbic acid (AA). The respective current peaks were clearly separated from each other, thus enabling selective detection of these compounds coexisting in a mixture. For the prepared sensor a linear relationship between the current response of EP electrooxidation at the potential of peak maximum (ip) and the concentration of EP in solution (cEP) in the presence of constant AA concentration was found in the broad range of cEP (R2 ≥ 0.9997, 1.0 × 10−8 M to 9.0 × 10−4 M) with a high detection limit (7.4 × 10−9 M), excellent reproducibility as well as high stability.

 Please wait while we load your content...
Please wait while we load your content...