Influence of a reagents addition strategy on the Fenton oxidation of rhodamine B: control of the competitive reaction of ·OH†
Abstract
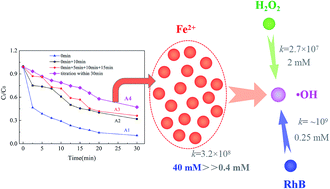
The Fenton system (Fe2+/H2O2) generates ·OH with a high oxidation potential. However, as reactants themselves, H2O2 and Fe2+ can act as ·OH initiators as well as ·OH scavengers, leading to the need for a high dosage of reactants and increased costs. As a mixing-sensitive reaction, the ·OH-related reaction kinetics (·OH with Fe2+, H2O2, and RhB) was determined from the reaction rates (which were a constant in this work) and stoichiometry, in which the latter could be regulated by an addition strategy of Fenton reagents. This suggests that ·OH competitive reactions could be controlled by applying a macrolevel addition strategy. Herein, the effects of different addition approaches of Fe2+ and H2O2 on ·OH competitive reactions were quantitatively and systematically studied by analyzing the removal of the model pollutant RhB. We found that without stirring, and compared with a one-time addition, once H2O2 or Fe2+ was added in a step-wise pattern (e.g., one drop by one drop, 2 times, or 4 times), a high concentration of H2O2 or Fe2+ existed in a localized place for a longer period, resulting in a lower proportion of ·OH reacting with RhB, which we ascribed to an enhanced reaction between Fe2+, H2O2, and ·OH. However, when H2O2 and Fe2+ were added from two close points without stirring, a larger proportion of ·OH was scavenged by H2O2 and Fe2+; while under stirring, even a one-time addition of H2O2 or Fe2+ could cause severe scavenging of ·OH. The results also revealed a linear relationship between the RhB removal percentage and wavelength blue-shifts. This study showed that microlevel ·OH competitive reactions could be controlled by applying a macrolevel addition strategy of Fenton reagents without the addition of external chemicals. The results suggest this methodology can also offer an approach to lower ·OH invalid consumption by regulating the addition strategy in bigger reactors.


 Please wait while we load your content...
Please wait while we load your content...