Synthesis of catalytically active gold clusters on the surface of Fe3O4@SiO2 nanoparticles†
Abstract
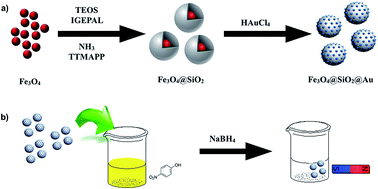
This work proposes a novel method to obtain catalytically active gold clusters by using the water-soluble 5,10,15,20-Tetrakis(4-trimethyl-ammonio-phenyl)porphyrin under mild conditions instead of using strong reducing agents. Uniform gold clusters were obtained with a diameter comprised between 1 and 2 nm and long-term stability. Furthermore, the water-soluble porphyrin was immobilized on the SiO2 shell of a core@shell Fe3O4@SiO2 nanoparticles to directly synthesize gold clusters onto the surface of Fe3O4@SiO2 nanoparticles featuring their magnetic recovery. Fe3O4@SiO2@Au nanoparticles were found to be catalytically active for the reduction of 4-nitrophenol to 4-aminophenol using NaBH4 and giving very high pseudo-first-order rate constant comprised between 0.7 and 2.7 min−1. Our results demonstrate that Fe3O4@SiO2@Au nanoparticles are stable catalysts and do not degrade during the catalytic process under the reaction conditions enabling their magnetic recuperation.


 Please wait while we load your content...
Please wait while we load your content...