Segregation of confined ionic liquids inducing the formation of super-micropores in the silica matrix†
Abstract
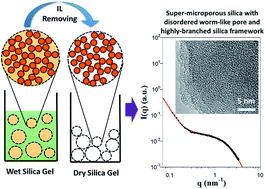
In the present work, we prepared a super-microporous silica gel via a nonhydrolytic methanol–formic-acid solvolytic sol–gel process and using a hydrophobic short-chain ionic liquid (IL), 1-butyl-3-methylimidazolium hexafluorophosphate, as the template solvent and tetraethyl orthosilicate (TEOS) as the precursor. Low-temperature solvent extraction (65–70 °C) and subsequent vacuum drying were adopted to remove the confined IL. Transmission electron microscopy (TEM), nitrogen sorption (BET), and small/wide-angle X-ray scattering (S/WAXS) experiments were carried out to explore the structures of the prepared porous silica gels and the corresponding formation mechanism. The results from BET and TEM revealed that the synthesized silica gels with higher IL/TEOS molar ratio of ≥2.0 formed fractal matrix with embedded disordered worm-like super-micropore (pore size of 1.1–1.2 nm and surface areas of 690–800 m2 g−1). Further, S/WAXS analysis revealed the formation of super-micropore in the silica matrix strongly depended on the formation of highly-branched fractal silica framework (mass-fractal dimension of silica matrix Df > 2.8) and the segregation of the confined IL, which was basically driven by the strong IL/pore wall repulsive interactions when excess amount of IL was used here.


 Please wait while we load your content...
Please wait while we load your content...