Simple synthesis of lithium-doped sulfated titania nanoparticles and their high visible light photocatalytic activity under negative bias electrostatic field
Abstract
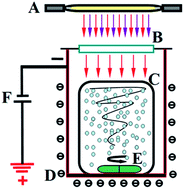
Li-doped TiO2/SO42− nanoparticles was successfully synthesized via a simple calcinination process in a vacuum environment using Ti(SO4)2 and LiBr as precursors, and were characterized by TEM, XRD, IR, DLS, XPS and UV-vis (DRS). Li doping can reduce the diameters of TiO2 nanoparticles and affect the surface chemical forms and structures. The as-prepared photocatalysts with different LiBr contents exhibited a notably enhanced degradation efficiency for methylene blue, and exhibited the highest photocatalytic activity under UV light when the molar doping ratio of Li was 0.0125. Moreover, the most photocatalytically efficient sample also showed much higher activity for eliminating methylene blue under visible light (380 < λ < 700 nm) by applying a negative bias than P25 and the control samples without applying a negative bias. The introduced negative bias electrostatic fields could not only render TiO2 responsive to visible light, but was also able to increase the lifetime of the photo-excited charges in the doped semiconductor. This provides a facile, fast and universal method to rapidly degrade organic materials based on the Franz–Keldysh effect and the synergetic effects of the electrostatic force.

 Please wait while we load your content...
Please wait while we load your content...