Nano-cuprous oxide catalyzed one-pot synthesis of a carbazole-based STAT3 inhibitor: a facile approach via intramolecular C–N bond formation reactions†
Abstract
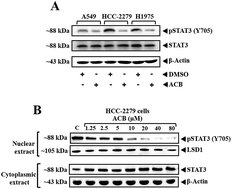
In this study, we report the one-pot synthesis of substituted carbazole derivatives using nano cuprous oxide as a catalyst via intramolecular C–N bond forming reactions. Among the synthesized carbazoles, 3′-((3-acetyl-6-chloro-9H-carbazol-9-yl)methyl)-[1,1′-biphenyl]-2-carbonitrile (ACB) was identified as a lead antiproliferative agent against lung cancer cell lines A549 and LLC with an IC50 of 13.6 and 16.4 μM respectively. Furthermore, we found that the lead compound suppresses the constitutive phosphorylation of STAT3 (Tyr-705) in A549, HCC-2279 and H1975 cells. We analyzed the levels of phospho-STAT3 and LSD1 in the nuclear extract of ACB treated HCC-2279 cells to evaluate the transcriptional activity of STAT3. We found the downregulation of phospho-STAT3 without any change in the expression of LSD1 indicating that ACB downregulates the transcriptional activity of STAT3. Molecular docking analysis revealed that ACB makes a favorable interaction with Arg-609 and Ser-613 in the pTyr site of the SH2 domain of STAT3.

 Please wait while we load your content...
Please wait while we load your content...