N-Doped and Cu-doped TiO2-B nanowires with enhanced photoelectrochemical activity†
Abstract
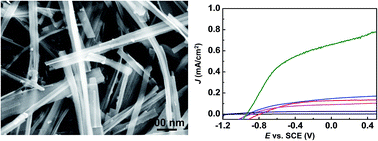
TiO2-B is a metastable phase of titania with an interesting crystal structure and electrochemical properties. N-Doped and Cu-doped TiO2-B nanowires (NWs) were synthesized by hydrothermal and microwave-assisted hydrothermal methods, respectively. All the NWs are single crystal with the same crystal orientation regardless of the synthesis method and doping situation. The doped NWs together with their corresponding pristine NWs were fabricated into a photoelectrochemical (PEC) anode for water oxidation and their photoactivity performances were studied and compared under different illumination wavelength ranges. The Cu-doped TiO2-B NWs exhibited a significantly higher photocurrent density than all the other samples under direct Xe lamp illumination; while their performance rapidly dropped below the hydrothermal NWs when the UV illumination was cut off. On the contrary, N-doped TiO2-B NWs exhibited significantly enhanced photoactivity particularly in the visible light range. X-ray photoelectron spectroscopy revealed that N-doping could narrow the electronic bandgap of TiO2, while the Cu-doping had little impact on the bandgap but rather improved the electrical conductivity. This research provides a new insight into elemental and phase control of TiO2-based solar energy harvesters.

 Please wait while we load your content...
Please wait while we load your content...