Two-dimensional nanostructures of non-layered ternary thiospinels and their bifunctional electrocatalytic properties for oxygen reduction and evolution: the case of CuCo2S4 nanosheets†
Abstract
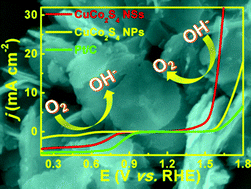
Two-dimensional (2D) transition metal chalcogenide nanostructures exhibit unique electronic, optoelectronic and mechanical properties, showing great potential for innovation of future electronics, renewable energy, sensing, and catalysis fields. Despite achieving great progress, fabricating 2D nanostructures of non-layered ternary thiospinels remains a great challenge, and their bifunctional electrocatalytic properties toward both oxygen reduction and evolution reactions (ORR and OER) have not been explored. In this paper, 2D nanostructures of an earth-abundant non-layered ternary thiospinel compound – CuCo2S4 nanosheets (NSs) – with their (111), (022) and (004) planes mainly exposed, are synthesized via a “leveling metal activity and structure-directed one-pot sulfurization” strategy. The electrocatalytic tests in alkaline solution show that such CuCo2S4 NSs can efficiently catalyze both the ORR and OER, whose bifunctional catalytic properties are superior to binary metal sulfide nanostructures, Pt/C, CuCo2S4 nanoparticles, and recently reported some bifunctional oxygen-electrode catalysts (e.g. CoxSy@C-1000, N-doped G/CNTs, Co3O4/NBGHSs, CoFe2O4, CoxMn3−xO4, MnOx, NiCo2S4 hollow spheres, NiCo2S4@N/S-rGO, etc.). Combined with spin-polarized density functional theory computations based on a computational hydrogen electrode model, their excellent bifunctional catalytic properties originate from the presence of the two special facets, (022) and (004), which have different preferences in terms of the ORR/OER. This work not only enriches the current 2D material family but also paves the way for rational design of advanced multifunctional 2D electrocatalysts for use in renewable energy fields.


 Please wait while we load your content...
Please wait while we load your content...