Intense blue emission and a reversible hypsochromic shift of luminescence caused by grinding based on silver(i) complexes†
Abstract
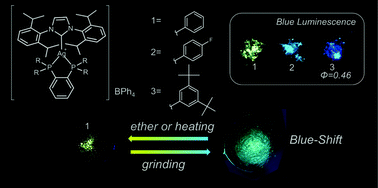
Silver(I) complexes bearing N-heterocyclic carbene and diphenylphosphinobenzene ligands, [Ag(IPr)(dppbz)]BPh4 (1, IPr = bis(2,6-diisopropylphenyl)imidazole-2-ylidene, dppbz = 1,2-bis(diphenylphosphino)benzene), [Ag(IPr)(dfpbz)]BPh4 (2, dfpbz = 1,2-bis[di(4-fluorophenyl)phosphino]benzene), and [Ag(IPr)(dtbpbz)]BPh4 (3, dtbpbz = 1,2-bis[bis[3,5-di(tert-butyl)phenyl]phosphino]benzene), were newly synthesized and characterized. These complexes, especially 3, exhibit intense blue emission in the solid state. The maximum wavelength and lifetime of the photoluminescence of 3 are 440 nm and 14 μs, respectively. The quantum yield of the photoluminescence of 3 in the solid state is 0.5, which is one of the highest values among blue-emissive silver(I) complexes ever reported. Introduction of bulky tert-butyl groups at 3- and 5-positions of the phenyl groups in the diphosphine ligand plays a key role in both the blue-shift and the enhancement of the photoluminescence. Additionally, the reversible hypsochromic shift of the long-lived emission caused by the grinding process is observed for one of the complexes.


 Please wait while we load your content...
Please wait while we load your content...