Titania hollow spheres modified with tungstophosphoric acid with enhanced visible light absorption for the photodegradation of 4-chlorophenol†
Abstract
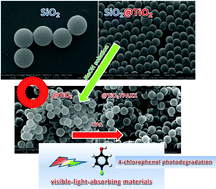
Titania hollow spheres were synthesized using silica nanospheres as the template. The core was removed using NaOH solution. They were subsequently impregnated with tungstophosphoric acid (TPA) solutions and annealed at two different temperatures (100 and 500 °C). These materials were characterized by several physicochemical techniques (XRD, BET, SEM, DRS, FT-IR, FT-Raman and 31P MAS-NMR). The 31P MAS-NMR and FT-IR characterization showed that the main species present in the samples was the [PW12O40]3− anion, which was partially transformed into the [P2W21O71]6− anion during the synthesis and drying step. 31P MAS-NMR, and FT-Raman characterization revealed the evidence of a strong interaction between the Keggin anion of TPA and TiO2 surfaces, possibly due to the formation of surface heteropolyacid-TiO2 complexes. The DRS results showed that the absorption threshold onset continuously shifted to the visible region with increased TPA concentration and calcination at 500 °C. The enhanced visible light absorption could be related to the formation of a surface complex TPA Keggin anion–TiO2. The catalytic activity of the materials in the photodegradation of 4-chlorophenol under UV and visible light irradiation increased when the TPA content and the calcination temperature of the samples were raised.
- This article is part of the themed collection: Solar chemistry & photocatalysis – environmental applications

 Please wait while we load your content...
Please wait while we load your content...