Single electron transfer promoted photoaddition reactions of α-trimethylsilyl substituted secondary N-alkylamines with fullerene C60†
Abstract
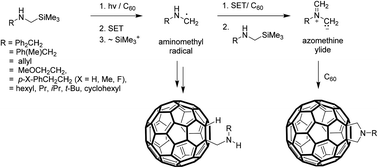
Single electron transfer (SET) promoted photoaddition reactions of secondary N-α-trimethylsilyl-N-alkylamines to C60 were explored to gain a deeper understanding of the mechanistic pathways followed and to expand the library of novel types of organofullerenes that can be generated using this approach. The results show that photoreactions of 10% EtOH–toluene solutions containing C60 and N-α-trimethylsilyl-N-alkylamines produce either aminomethyl-1,2-dihydrofullerenes or symmetric fulleropyrrolidines as major products depending on the nature of alkyl substituents. In contrast, photoreactions of 10% EtOH–ODCB solutions of these amines with C60 mainly lead to the formation of symmetric fulleropyrrolidines. Based on the analysis of product distributions and the results of earlier studies, two feasible mechanistic pathways are proposed for these processes. One route is initiated by SET from the amine substrates to the triplet-excited state of C60 to form the corresponding aminium radicals and C60 anion radicals. EtOH-promoted desilylation of the aminium radicals then takes place to produce aminomethyl radicals which can either add to C60 or couple with the C60 radical anions to form respective radicals or anion precursors of aminomethyl-1,2-dihydrofullerene products. The competing pathway leading to the generation of symmetric fulleropyrrolidines also involves the formation of aminomethyl radicals by using the sequential SET-desilylation process. In this route, the aminomethyl radicals are oxidized by SET to C60 to form iminium ions, which are then transformed to azomethine ylides by a pathway involving a second molecule of the secondary amine. Dipolar cycloaddition of the azomethine ylides to C60 forms the symmetric fulleropyrrolidine cycloadducts. Importantly, the observation that symmetric fulleropyrrolidines are the sole products formed in photoreactions between N-α-trimethylsilyl-N-alkylamines and C60 in 10% EtOH–ODCB has synthetic significance.

 Please wait while we load your content...
Please wait while we load your content...