Helical α/β-depsipeptides with alternating residue types: conformational change from the 11-helix to the 14/15-helix†
Abstract
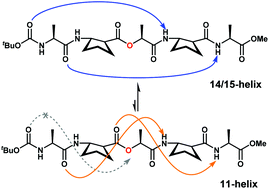
Short α/β-peptides that consist of alternating L-α-amino acids and trans-2-aminocyclopentanecarboxylic acid are known to adopt both 11- and 14/15-helical conformations in solution. We report short α/β-depsipeptides containing (S)-lactic acid as the third residue from the N-terminus. The α/β-depsipeptide pentamers and heptamers adopt 14/15-helical conformations analogous to the α-helix in the crystal state and display 14/15-helical conformations predominantly in solution.

 Please wait while we load your content...
Please wait while we load your content...