Diacetoxyiodobenzene assisted C–O bond formation via sequential acylation and deacylation process: synthesis of benzoxazole amides and their mechanistic study by DFT†
Abstract
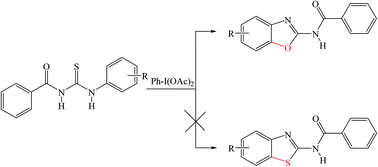
An efficient method for the transformation of N-substituted-N′-benzoylthioureas to substituted N-benzoxazol-2-yl-amides using diacetoxyiodobenzene (DIB) is described in this work. The transformation follows the C–O bond formation leading to the benzoxazole derivative, due to oxidative dehydrogenation by DIB, instead of the expected C–S bond formation of the benzothiazole moiety. The C–O bond formation leading to benzoxazole is due to consecutive acylation and deacylation in conjunction with the reduction of two moles of DIB. A plausible mechanism was proposed for the reaction and density functional calculations were also performed to study the reaction mechanism.

 Please wait while we load your content...
Please wait while we load your content...