Total synthesis of (±)-epithuriferic acid methyl ester via Diels–Alder reaction†
Abstract
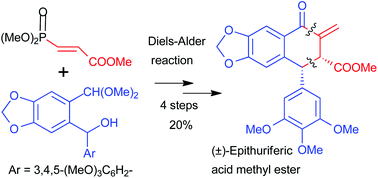
In this paper, we have described the first total synthesis of (±)-epithuriferic acid methyl ester from non-natural sources, in four steps (20% overall yield). The key step involves the Diels–Alder reaction of isobenzofuran with methyl 3-(dimethoxyphosphoryl)acrylate which is controlled by “ortho” regio- and endo stereoselectivities due to the COOMe group.

 Please wait while we load your content...
Please wait while we load your content...