Targeted delivery of doxorubicin to tumour tissues by a novel legumain sensitive polygonal nanogel†
Abstract
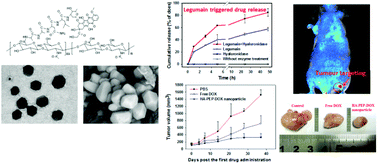
Targeted delivery of cytotoxic drugs to tumour tissue has great importance for successful chemotherapy. Legumain is an asparaginyl endopeptidase that is highly up-regulated in a number of solid tumours. The aim of this work was to prepare a novel hyaluronic acid (HA) based legumain sensitive nanogel for the delivery of doxorubicin with a high targeting efficiency both in vitro and in vivo. The legumain sensitive property is achieved by the conjugation of doxorubicin with HA via a legumain substrate peptide bridge. This HA derivative is further crosslinked in a water/oil solvent system to form a polygonal nanogel. Doxorubicin released in the tumour tissue is sustained thanks to the combined action of legumain and hyaluronidase, which are both overexpressed in tumour tissues. Hyaluronic acid could act as a targeting agent to CD44 (HA receptor), which further improved the in vivo target effect and enhanced in vitro cellular uptake. The developed nanogel exhibited a high therapeutic index that improved tumour inhibition effects and reduced system toxicity in a lung cancer mice model. These results highlighted the advantages of using this multi-functional material for a successful delivery of doxorubicin against cancer.

 Please wait while we load your content...
Please wait while we load your content...