Chemical synthesis of Nd2Fe14B hard phase magnetic nanoparticles with an enhanced coercivity value: effect of CaH2 amount on the magnetic properties†
Abstract
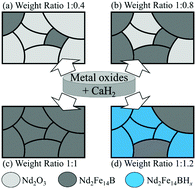
Nd2Fe14B hard phase magnetic nanoparticles were successfully synthesized using a chemical synthesis route followed by a reduction and diffusion process without consuming a large amount of energy. Nd(acac)3·xH2O, Fe(acac)3 and (C2H5)3NBH3 were used as precursors and CaH2 was used as a reducing agent for the reduction and diffusion process. The crystal structure and composition of the resultant Nd–Fe–B nanoparticles were determined using X-ray diffraction (XRD) and energy dispersive X-ray spectroscopy (EDX) measurements. The overall morphology and magnetic properties of the final products were investigated using transmission electron microscopy (TEM) and vibrating sample magnetometry (VSM) at 300 K. The effect of the relative quantity of CaH2 on the magnetic properties of the final products was studied in detail with different weight ratios of CaH2 to the preliminary reduced metal oxide powders, using high resolution TEM (HRTEM), XRD and VSM measurements. The different mechanisms of the reduction and diffusion process with different amounts of CaH2 were comparatively studied. An insufficient amount of CaH2 resulted in the presence of residual unreduced metal oxide powders, decreasing the magnetic coercivity. On the other hand, an excessive amount of CaH2 caused H2 gas to be evolved during the washing process with water, resulting in hydrogenation of the Nd2Fe14B to produce Nd2Fe14BHx. The relative amount of CaH2 had a critical effect on the magnetic properties of the prepared Nd2Fe14B-nanoparticles.

 Please wait while we load your content...
Please wait while we load your content...