Replacing bisphenol-A with bisguaiacol-F to synthesize polybenzoxazines for a pollution-free environment†
Abstract
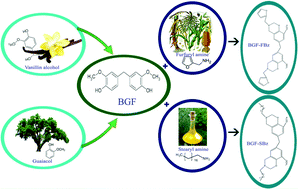
In recent years, bisguaiacol F (BGF) is considered as a green alternative to bisphenol A (BPA). The present work deals with the synthesis of BGF and BGF-based benzoxazines, viz., bis[(3-(furan-2-yl) methyl)-3,4-dihydro-2H benzoxazin-6-yl]methane and bis[(3-octadecyl)-3,4-dihydro-2H benzoxazin-6-yl]methane, as an alternative to BPA and polybenzoxazines derived from BPA. A solventless method was adopted for the synthesis of BGF and the benzoxazine monomers. The structure and purity of BGF is confirmed by FT-IR, NMR (1H and 13C), MS and GC. The monomers were polymerized via ring opening polymerization by heating as shown by FT-IR (disappearance of the peak due to the oxazine ring at 933 cm−1). Differential scanning calorimetric (DSC) analysis of the monomers shows a wide range of processability between 77 and 122 °C. Thermal properties of the furfurylamine based polybenzoxazine (BGF-FPbz) and stearylamine based polybenzoxazine (BGF-SPbz) were investigated by thermogravimetric analysis (TGA). The results indicate that BGF-FPbz shows the maximum thermal stability when compared with BGF-SPbz. Both the polymers exhibit good flame retardant properties as shown by their LOI values (more than 26).

 Please wait while we load your content...
Please wait while we load your content...