One-pot synthesis and application of novel amino-functionalized silica nanoparticles using guanidine as amino group†
Abstract
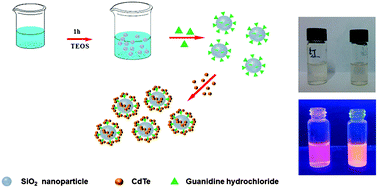
We developed a simple and efficient strategy to prepare monodisperse amino-functionalized silica nanoparticles (SiNPs). Guanidine hydrochloride was selected as an amination reagent because it can be easily absorbed by SiNPs through charge interaction. The reverse microemulsion method was employed to synthesize guanidine-modified silica nanoparticles (SiNPs) in a one-pot procedure. The obtained SiNPs were then characterized through FTIR, SEM, and TEM. The nitrogen content on SiNP surfaces was directly measured by using an elemental analyzer. Results revealed that the amino group concentration on the SiNP surfaces was 1.5 × 10−6 mol mg−1. The monodispersity of the amino-functionalized SiNPs prepared through this method and the concentration of their amino groups were higher than those obtained via traditional methods. As such, these SiNPs were utilized to fix quantum dots on the SiNP surfaces. The guanidine-modified SiNPs coated with QDs were further characterized through SEM, TEM, and fluorescence spectroscopy. Furthermore, the modified SiNPs as basic nanocatalysts were used to catalyze the Henry reaction. Conversion was high under optimal conditions, and reaction time was remarkably shortened. The nanocatalysts could also be recycled for at least four additional cycles after they were centrifuged, washed with ethanol, air-dried, and immediately reused. Therefore, guanidine hydrochloride is a feasible amination reagent, and guanidine-functionalized SiNPs are promising materials in many fields because of their advantages.

 Please wait while we load your content...
Please wait while we load your content...