Can fused-pyrrole rings act as better π-spacer units than fused-thiophene in dye-sensitized solar cells? A computational study†
Abstract
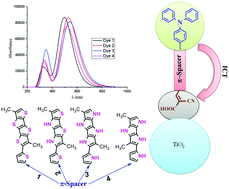
Fused-pyrrole spacer groups have been exploited for the first time computationally to improve the efficiency of dye-sensitized solar cells (DSSCs). Known dye 1 (TPA-TTAR-T-A) containing tetrathienoacene as a spacer group has achieved 10% efficiency, which is comparable to that of ruthenium complex dyes. The DFT study reveals that fused-pyrrole as a spacer in dye 3 can augment the efficiency of DSSCs and can perform better than dye 1. The pyrrole ring possesses lower resonance energy (21.0 kcal mol−1) than the commonly used thiophene (29–34 kcal mol−1) spacers. The higher conjugative effect of fused-pyrrole rings influence the short-circuit current density (Jsc), including driving force of electrons (ΔGinjection) and singlet excited state lifetime (τ). Further, open-circuit photovoltage (Voc), which depends on dipole moments (μnormal) and number of electrons transferred from the dye to the TiO2 surface (Δq), are also influenced with such conjugative effects. The calculated λmax (539 nm) and ΔGinjection (−1.82 eV) of dye 3 is much higher than the dye 1 (λmax = 497 nm and ΔGinjection = −1.28 eV). The calculated results show that the singlet excited state lifetime of dye 3 (2.2 ns) is higher than the dye 1 (1.7 ns). Dye 3 should have better Jsc value than 1 as the obtained ΔGinjection, τ and λmax values are higher in the former case. The DFT calculations also suggest that the open-circuit photovoltage (Voc) of 3 would be higher than 1 and hence can augment the efficiency of the designed dye 3. The study was extended with tetrapyrrolobenzothiadiazole spacer groups (dyes 6 and 8) which are also shown to be promising candidates to improve the efficiencies of DSSCs. Importantly, the tendency of aggregation of dyes can also be prevented by introducing bulky groups in the fused-pyrrole ring which can also help to enhance the efficiency of designed dyes relative to reported dyes. The exploration of novel sensitizers with fused-pyrrole spacer groups would provide potential candidates for DSSCs.


 Please wait while we load your content...
Please wait while we load your content...