A novel fluorene based “turn on” fluorescent sensor for the determination of zinc and cadmium: experimental and theoretical studies along with live cell imaging†
Abstract
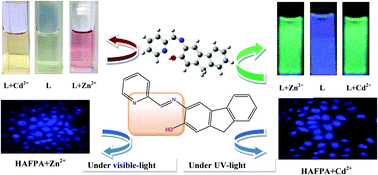
A new fluorene–pyridine linked imine conjugate, HAFPA, has been synthesized and characterized. HAFPA has been successfully found to recognize Zn2+ and Cd2+ selectively among a wide range of biologically relevant metal ions. The chemosensing behaviour of the ligand has been demonstrated through 1H NMR, ESI-MS, emission, absorption, colour change, luminescence spectra. The probe HAFPA displays a prominent fluorescence enhancement along with a large red shift (∼60 nm and 65 nm), which may be because of the CHEF (chelation-enhanced fluorescence) processes after its interaction with Zn2+ and Cd2+ respectively. The binding of the ligand with Zn2+ and Cd2+ was investigated through ESI-MS, Job's plot and reversibility studies. The fluorescence property of the ligand after complexation with Zn2+ and Cd2+ can be restored in the presence of EDTA and sulphide ions. This reversibility phenomenon is utilized in the construction of a logic gate. The DFT, TDDFT and triplet excited state calculations were performed in order to demonstrate the structure of the ligand and its complexes and their electronic properties. Importantly, our chemosensor could be used to detect intracellular Zn2+ and Cd2+ with no cytotoxicity in A549 cells, and a colorimetric test strip of the ligand for the detection of both the ions could be useful for all practical purposes.

 Please wait while we load your content...
Please wait while we load your content...