Investigating the DMPO-formate spin trapping method for the study of paper iron gall ink corrosion
Abstract
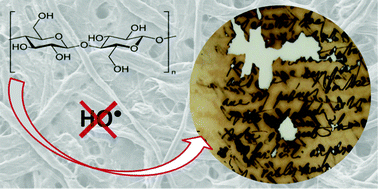
Paper degradation by iron gall ink is a challenging issue for the conservation of ancient manuscripts. It is usually attributed to both FeII catalyzed oxidation and acid catalyzed hydrolysis of cellulose, yet the dominant degradation pathway remains an open question. In this work, electron spin resonance spectroscopy (ESR) was used to identify radicals involved in cellulose degradation. Papers impregnated with iron salts were put in contact with an aqueous solution of DMPO (spin-trap) and analyzed by ESR. No DMPO–OH˙ adducts were detected while the addition of formate, an intermediary target molecule, led on the contrary to the detection of a significant amount of DMPO–CO2˙− adducts. This reveals the predominant occurrence of reactive oxygen species (ROS) other than hydroxyl radicals (most likely superoxide or hydroperoxyl radicals). The production of DMPO–CO2˙− adducts was found to be oxygen dependent and to originate mainly from ferrous ions lixiviated from the impregnated papers. At high iron loads (above 1 mmol L−1), it was shown that quantification of these adducts should be done with great care because of a competing formation of hydroxylamine that jeopardizes the ESR signal. Complementary TPA hydroxylation measurements performed by HPLC on ink and iron sulfate impregnated papers revealed some production of hydroxyl radicals but at a concentration several orders of magnitude below that of the ROS detected with formation of DMPO–CO2˙− adducts. This HO˙ production was moreover not correlated with cellulose chain scissions, indicating that autooxidation by these radicals is not the main mechanism of cellulose depolymerisation in presence of iron gall ink.


 Please wait while we load your content...
Please wait while we load your content...