Label-free and dual-amplified electrochemical detection of Hg2+ based on self-assembled DNA nanostructures and target-triggered exonuclease cleavage activity
Abstract
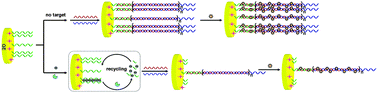
In this work, a DNA nanostructure-enabled electrochemical biosensor was developed for sensitive Hg2+ detection via Hg2+-tiggered exonuclease III (Exo III) cleavage amplification. In the absence of Hg2+, long-range self-assembly of the capture probe (CP) and two auxiliary strands in hybridization chain reaction (HCR) constructed the functional interfaces of the electrode. The formed DNA nanostructures acted as carriers of the signaling molecule ([Ru(NH3)6]3+), which resulted in a remarkable electrochemical signal output. The presence of Hg2+ led to the formation of a duplex-like DNA structure by two adjacent T-rich CPs through specific T–Hg2+–T coordination chemistry, triggering the cleavage activity of Exo III. Exo III-assisted target recycling led to the reduction of CPs and restrained the extension of the DNA nanostructure. In which, less absorbed [Ru(NH3)6]3+ led to the decrease of the electrochemical signal. The HCR and Exo III cleavage amplification strategy significantly improved the sensitivity of the proposed biosensor with a detection limit as low as 0.14 pM. Moreover, this strategy could be extended to sensitive and selective detection of a wide range of targets.

 Please wait while we load your content...
Please wait while we load your content...