Investigation of uranium interactions with calcium phosphate-binding proteins using ICP/MS and CE-ICP/MS
Abstract
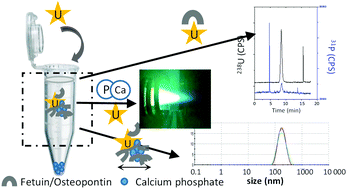
During long-term exposure, uranium accumulates in bone. Since uranium in U(VI) complexes shares similar coordination properties to calcium, this toxicant is assumed to be exchanged with calcium ions at the surfaces of bone mineral crystals. Recently, two proteins involved in bone turnover, fetuin A and osteopontin, were shown to exhibit a high affinity for U(VI). A common biochemical feature of both fetuin A and osteopontin is their inhibiting role in calcium phosphate precipitation. Therefore it is conceivable that complexation of U(VI) with these proteins may alter their interaction with calcium and/or calcium phosphate. Quantitative analyses of calcium, phosphorus and uranium performed using inductively coupled plasma/mass spectrometry (ICP/MS) demonstrated the inhibition of the precipitation of calcium phosphate by fetuin A and osteopontin for 2 h. In addition, the presence of U(VI) did not seem to alter the duration of this process. However, dynamic light scattering studies revealed that the size of the colloidal particles formed with osteopontin was altered by the presence of U(VI) in a concentration-dependent manner. Finally, using hyphenated capillary electrophoresis-ICP/MS (CE-ICP/MS), we showed that in these systems, at a low concentration of U(VI) (protein : U(VI) 8 : 1), U(VI) might remain in solution by forming a complex with proteins and not by sequestration of a precipitate of either autunite or uranyl orthophosphate.

 Please wait while we load your content...
Please wait while we load your content...