Non-trivial network driven modifications of ion transport in an ionic liquid confined inside a polymer system†
Abstract
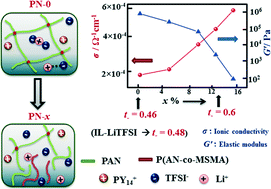
We discuss here the polymer specificities which completely govern in a non-trivial manner the effective ion transport in polymer gel electrolytes, an important class of soft matter electrolytes. Confinement of a lithium bis(trifluoromethanesulfonyl)imide (LiTFSI)–pyrrolidinium-cation based ionic liquid solution inside a polymer physical network composed of two different polymers with different functionalities is achieved. A physical network of two polymers with different functional groups, viz., one with acrylate and another with acrylonitrile (PAN) leads to multiple interesting consequences. Due to chemical differences between the acrylate-based polymer (formed from (3-trimethoxysilyl)propyl methacrylate (MSMA) monomers) and PAN, the physical knots in the PAN network unlock, leading to a decrease in the elastic modulus with improved mechanical compliance and chain flexibility in the gel. Additionally, ion–polymer interactions increase, resulting in higher free charge carrier density in the gel compared to the unconfined ionic liquid solution. Thus, ion transport is no longer assisted by the ionic liquid and polymer relaxations, as it would be in conventional polymer electrolytes, but fully driven by the chemical characteristics of the polymer physical network. Notably, the polymer matrix significantly influences the anion mobility and transference number. Contrary to the unconfined ionic liquid electrolyte where cations (predominantly due to the pyrrolidinium cation) and anions contribute to the ionic conductivity, the ionic conductivity in PN gels is predominantly due to anions. The gel ionic conductivity is nearly half an order of magnitude higher than that of the unconfined ionic liquid electrolyte and displays good dimensional stability and electrochemical performance in a separator-free battery configuration. Most importantly, this work may revitalize research on single-ion conductors and stimulate new and simple chemical designs of polymer electrolytes displaying high single-ion conductivity.

 Please wait while we load your content...
Please wait while we load your content...