Stiffness of the C-terminal disordered linker affects the geometry of the active site in endoglucanase Cel8A†
Abstract
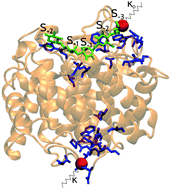
Cellulosomes are complex multi-enzyme machineries which efficiently degrade plant cell-wall polysaccharides. The multiple domains of the cellulosome proteins are often tethered together by intrinsically disordered regions. The properties and functions of these disordered linkers are not well understood. In this work, we study endoglucanase Cel8A, which is a relevant enzymatic component of the cellulosomes of Clostridium thermocellum. We use both all-atom and coarse-grained simulations to investigate how the conformations of the catalytic domain of Cel8A are affected by the disordered linker at its C terminus. We find that when the endoglucanase is bound to its substrate, the effective stiffness of the linker can influence the distances between groups of amino-acid residues throughout the entire enzymatic domain. In particular, variations in the linker stiffness can lead to small changes in the geometry of the active-site cleft. We suggest that such geometrical changes may have an effect on the catalytic activity of the enzyme.

 Please wait while we load your content...
Please wait while we load your content...