Integrated machine learning, molecular docking and 3D-QSAR based approach for identification of potential inhibitors of trypanosomal N-myristoyltransferase†
Abstract
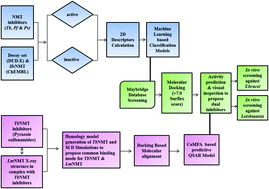
N-Myristoyltransferase (NMT) catalyzes the transfer of myristate to the amino-terminal glycine of a subset of proteins, a co-translational modification involved in trafficking substrate proteins to membrane locations, stabilization and protein–protein interactions. It is a studied and validated pre-clinical drug target for fungal and parasitic infections. In the present study, a machine learning approach, docking studies and CoMFA analysis have been integrated with the objective of translation of knowledge into a pipelined workflow towards the identification of putative hits through the screening of large compound libraries. In the proposed pipeline, the reported parasitic NMT inhibitors have been used to develop predictive machine learning classification models. Simultaneously, a TbNMT complex model was generated to establish the relationship between the binding mode of the inhibitors for LmNMT and TbNMT through molecular dynamics simulation studies. A 3D-QSAR model was developed and used to predict the activity of the proposed hits in the subsequent step. The hits classified as active based on the machine learning model were assessed as the potential anti-trypanosomal NMT inhibitors through molecular docking studies, predicted activity using a QSAR model and visual inspection. In the final step, the proposed pipeline was validated through in vitro experiments. A total of seven hits have been proposed and tested in vitro for evaluation of dual inhibitory activity against Leishmania donovani and Trypanosoma brucei. Out of these five compounds showed significant inhibition against both of the organisms. The common topmost active compound SEW04173 belongs to a pyrazole carboxylate scaffold and is anticipated to enrich the chemical space with enhanced potency through optimization.

 Please wait while we load your content...
Please wait while we load your content...