An integrated quartz tube atom trap coupled with solid sampling electrothermal vapourization and its application to detect trace lead in food samples by atomic fluorescence spectrometry†
Abstract
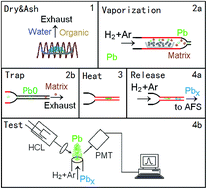
A two-stage diameter-varied quartz tube (QT) consisting of a QT electrothermal vapourizer, a quartz tube atom trap (QTAT) and two separate Ni–Cr electrical heating coils was interfaced with atomic fluorescence spectrometry (AFS). This is the first time that an atom trap is employed to trap Pb using electrothermal vapourization (ETV) as a solid sampling approach. The optimum ashing, vapourization, trapping, and releasing procedures and carrier gas were investigated. Under the optimized conditions, Pb in the ashed sample residues was vapourized at 850 °C, followed by analyte separation from the matrix with a 400 mL min−1 carrier gas of 10% H2/Ar (v/v) mixture through trapping Pb on the inner surface of the QTAT without heating. Finally, Pb was released from the QTAT by heating to 800 °C with the carrier gas switch-off and was then swept by the carrier gas into the AFS for measurement. The method LOD and the RSD of repeated measurements were 2 pg and 1.5%, respectively, indicating sufficient analytical sensitivity and precision. The Pb concentrations measured in reference materials by the proposed QT-ETV-QTAT-AFS method were within the certified values, and the spiked recoveries ranged from 95% to 106%, which demonstrated satisfactory accuracy for rapid and on-site determination of Pb in food samples. Furthermore, the vapourization, trapping and releasing mechanisms for Pb by QT-ETV-QTAT-AFS were also studied via tungsten coil ETV-QTAT-AFS, in situ atomic absorption spectrometry, X-ray photoelectron spectroscopy and other means. We deduced that the formation of atom clusters played a crucial role in Pb transportation to the detector.


 Please wait while we load your content...
Please wait while we load your content...