Speciation analysis of silver sulfide nanoparticles in environmental waters by magnetic solid-phase extraction coupled with ICP-MS†
Abstract
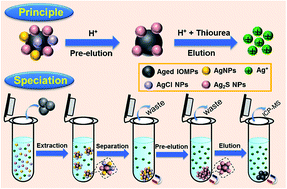
The growing production and widespread application of silver nanoparticles (AgNPs) have led to their release into the environment, where they are mostly transformed to silver sulfide nanoparticles (Ag2S NPs). Thus, speciation analysis of Ag2S NPs in environmental matrices is essential for understanding the environmental process and toxic effects of AgNPs. Herein, we report the use of aged iron oxide magnetic particles (IOMPs) as magnetic solid-phase extraction adsorbents for speciation analysis of Ag2S NPs. It was found that IOMPs are excellent adsorbents for the selective extraction of silver-containing nanoparticles (AgCNPs) including Ag2S NPs, AgNPs and AgCl NPs in the presence of Ag+. More importantly, Ag2S NPs can be distinguished from the other AgCNPs by sequential elution. After pre-eluting AgNPs and AgCl NPs as Ag+ with 2% (v/v) acetic acid and IOMPs as the sacrificial oxidants, Ag2S NPs were completely eluted as a Ag(I) complex by 10 mM thiourea in 2% (v/v) acetic acid and quantified directly by inductively coupled plasma mass spectrometry (ICP-MS). While the extraction of Ag+ was negligible, the maximum extraction of AgCNPs by IOMPs was attained at pH 4.9–6.2, and the interference of humic acid in the AgCNP extraction can be efficiently eliminated by adding Ca2+. Under optimized conditions, the method provides low detection limit (0.068 μg L−1) and high reproducibility (relative standard deviations < 5.1%) for Ag2S NPs. By simultaneously spiking 0.16–10.3 μg L−1 AgNPs and 0.53–14.8 μg L−1 Ag2S NPs, the Ag2S NP recoveries were in the range of 69.6–100.2% for the tap, river and lake waters; and in the range of 106.2–149.9% for the WWTP effluent due to the part sulfidation of AgNPs to Ag2S NPs. Our method is valid for the speciation analysis of Ag2S NPs in water samples, which provides an efficient approach for studying the sulfidation of AgNPs and Ag+.

 Please wait while we load your content...
Please wait while we load your content...