Homogeneous vinyl ester-based synthesis of different cellulose derivatives in 1-ethyl-3-methyl-imidazolium acetate†
Abstract
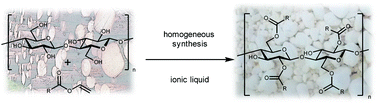
A homogeneous acylation of cellulose with different vinyl esters in the biodegradable and less toxic ionic liquid 1-ethyl-3-methyl-imidazolium acetate ([EMIM]OAc) is described for the first time. The reaction proceeds in the absence of any additional catalyst and glucose- and cellulose-esters with chain lengths of C8 to C16 are accessible by using equimolar amounts of acyl donor. Cellulose esters with a degree of substitution (DS) in the range of 0.9–3.0 were synthesised successfully. Different reaction parameters like reaction time, temperature and amount of substrate were systematically changed and analysed by NMR, IR and HPLC-GPC. The highest DS was achieved at 80 °C and a reaction time of 2 hours. Taking into consideration the literature, the DS and degree of polymerisation (DP) of fatty acid chloride and vinyl ester-based synthesis routes were compared. Similar DS-values were obtained, but the DP was significantly reduced during the synthesis using fatty acid chlorides in [BMIM]Cl. As an undesirable side reaction, acetates from [EMIM]OAc are bound to the cellulose backbone. The quantity of bound acetate groups during vinyl ester-based synthesis rose with decreasing polarity of the substrates but overall proved to be much lower compared to the literature described anhydride or fatty acid chloride based synthesis routes in [EMIM]OAc. This novel process was extended by using further acyl donors like vinyl benzoate, pivalate and 2-ethylhexanoate to demonstrate the applicability of the vinyl ester-based cellulose modification in [EMIM]OAc. [EMIM]OAc was recycled with an efficiency of ∼90% and reused for subsequent syntheses.

 Please wait while we load your content...
Please wait while we load your content...